B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system that does no work but that receives heat from the surroundings has
A) q < 0, ΔE > 0.
B) q > 0, ΔE < 0.
C) q = ΔE.
D) q = -ΔE.
E) w = ΔE.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Benzene is a starting material in the synthesis of nylon fibers and polystyrene (styrofoam) . Its specific heat capacity is 1.74 J/(g·K) . If 16.7 kJ of energy is absorbed by a 225-g sample of benzene at 20.0°C, what is its final temperature?
A) -22.7°C
B) 36.7°C
C) 42.7°C
D) 62.7°C
E) None of these choices are correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system initially has an internal energy E of 501 J. It undergoes a process during which it releases 111 J of heat energy to the surroundings, and does work of 222 J. What is the final energy of the system, in J?
A) 168 J
B) 390 J
C) 612 J
D) 834 J
E) This cannot be calculated without more information.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
For all processes, both q and w will have the same sign.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system that undergoes an adiabatic change and has work done on it by the surroundings has
A) w = ΔE.
B) w = -ΔE.
C) w > 0, ΔE < 0.
D) w < 0, ΔE > 0.
E) w > ΔE.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the ΔH°rxn for the decomposition of calcium carbonate to calcium oxide and carbon dioxide. ΔH°f [CaCO3(s) ] = -1206.9 kJ/mol; ΔH°f [CaO(s) ] = -635.1 kJ/mol; ΔH°f [CO2(g) ] = -393.5 kJ/mol CaCO3(s) → CaO(s) + CO2(g)
A) -2235.5 kJ
B) -1448.5 kJ
C) -178.3 kJ
D) 178.3 kJ
E) 2235.5 kJ
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system delivers 1275 J of heat while the surroundings perform 855 J of work on it. Calculate ΔE in J.
A) -2130 J
B) -420 J
C) 420 J
D) 2130 J
E) -1275 J
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
ΔH does not depend on the path of a reaction, but ΔE does.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Stoichiometric amounts of nitrogen gas and hydrogen gas react in a calorimeter to produce 5.00 g of ammonia gas. The calorimeter temperature rises 0.42°C. The calorimeter and water have a combined heat capacity of 32.16 kJ/K. Calculate the heat of formation of ammonia, ΔHf°, in kJ/mol. The formation reaction for ammonia is: 0.5N2(g) + 1.5H2(g) → NH3(g) .
A) -46 kJ/mol
B) -13.5 kJ/mol
C) -3.97 kJ/mol
D) 3.97 kJ/mol
E) 13.5 kJ/mol
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
40.0 g of ice cubes at 0.0°C are combined with 150. g of liquid water at 20.0°C in a coffee cup calorimeter. Calculate the final temperature reached, assuming no heat loss or gain from the surroundings. (Data: specific heat capacity of H2O(l) , c = 4.18 J/g·°C; H2O(s) → H2O(l) ΔH = 6.02 kJ/mol)
A) 0.0
B) 10.6
C) 30.7
D) 43.2
E) 56.4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sand is converted to pure silicon in a three step process. The third step is SiCl4(g) + 2Mg(s) → 2MgCl2(s) + Si(s) ΔH = -625.6 kJ What is the enthalpy change when 25.0 mol of silicon tetrachloride is converted to elemental silicon?
A) -25.0 kJ
B) -7820 kJ
C) -1.56 × 104 kJ
D) -3.13 × 104 kJ
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate q when 28.6 g of water is heated from 22.0°C to 78.3°C.
A) 0.385 kJ
B) 1.61 kJ
C) 6.74 kJ
D) 9.37 kJ
E) 1.61 × 103 kJ
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cold packs, whose temperatures are lowered when ammonium nitrate dissolves in water, are carried by athletic trainers when transporting ice is not possible. Which of the following is true of this reaction?
A) ΔH < 0, process is exothermic
B) ΔH > 0, process is exothermic
C) ΔH < 0, process is endothermic
D) ΔH > 0, process is endothermic
E) ΔH = 0, since cold packs are sealed
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The dissolution of barium hydroxide in water is an exothermic process. Which of the following statements is correct?
A) The enthalpy of solid barium hydroxide plus pure water is less than that of the solution, at the same temperature.
B) The enthalpy of solid barium hydroxide plus pure water is greater than that of the solution, at the same temperature.
C) The enthalpy of solid barium hydroxide plus pure water is the same as that of the solution, at the same temperature.
D) The temperature of the solution is lower than of the barium hydroxide and water before mixing.
E) When barium hydroxide dissolves in water, the system does work on the surroundings.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system receives 575 J of heat and delivers 425 J of work. Calculate the change in the internal energy, ΔE, of the system.
A) -150 J
B) 150 J
C) -l000 J
D) 1000 J
E) 575 J
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The specific heat capacity c of a metal is approximately related to its molar mass 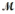 as follows: c ×
as follows: c × 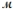 = 3R, where R is the universal gas constant, 8.314 J/mol·K. Use this relationship to identify the metal which has a specific heat capacity of 0.900 J/g·K.
= 3R, where R is the universal gas constant, 8.314 J/mol·K. Use this relationship to identify the metal which has a specific heat capacity of 0.900 J/g·K.
A) Li
B) Sn
C) Ca
D) Al
E) U
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system contracts from an initial volume of 15.0 L to a final volume of 10.0 L under a constant external pressure of 0.800 atm. The value of w, in J, is
A) -4.0 J.
B) 4.0 J.
C) -405 J.
D) 405 J.
E) 4.05 × 103 J.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An ideal gas (the system) is contained in a flexible balloon at a pressure of 1 atm and is initially at a temperature of 20.°C. The surrounding air is at the same pressure, but its temperature is 25°C. When the system has equilibrated with its surroundings, both systems and surroundings are at 25°C and 1 atm. In changing from the initial to the final state, which one of the following relationships regarding the system is correct?
A) ΔE < 0
B) ΔE = 0
C) ΔH = 0
D) w > 0
E) q > 0
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium hydroxide, which reacts with carbon dioxide to form calcium carbonate, was used by the ancient Romans as mortar in stone structures. The reaction for this process is Ca(OH) 2(s) + CO2(g) → CaCO3(s) + H2O(g) ΔH = -69.1 kJ What is the enthalpy change if 3.8 mol of calcium carbonate is formed?
A) -18 kJ
B) -69 kJ
C) -73 kJ
D) -260 kJ
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 72
Related Exams