A) Limiting reactant is Mg; 67 g of FeCl3 remain.
B) Limiting reactant is Mg; 134 g of FeCl3 remain.
C) Limiting reactant is Mg; 104 g of FeCl3 remain.
D) Limiting reactant is FeCl3; 2 g of Mg remain.
E) Limiting reactant is FeCl3; 87 g of Mg remain.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In combustion analysis, the carbon and hydrogen contents of a substance are determined from the CO2 and H2O, respectively, which are collected in the absorbers.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many atoms are in a drop of mercury that has a diameter of 1.0 mm? (Volume of a sphere is 4πr3/3; density of mercury = 13.6 g/cm3)
A) 2.1 × 1019
B) 1.7 × 1020
C) 2.1 × 1022
D) 1.7 × 1023
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Terephthalic acid, used in the production of polyester fibers and films, is composed of carbon, hydrogen, and oxygen. When 0.6943 g of terephthalic acid was subjected to combustion analysis it produced 1.471 g CO2 and 0.226 g H2O. If its molar mass is between 158 and 167 g/mol, what is its molecular formula?
A) C4H6O7
B) C6H8O5
C) C7H12O4
D) C4H3O2
E) C8H6O4
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Methanol (CH4O) is converted to bromomethane (CH3Br) as follows: CH4O + HBr → CH3Br + H2O If 12.23 g of bromomethane are produced when 5.00 g of methanol is reacted with excess HBr, what is the percentage yield?
A) 40.9%
B) 82.6%
C) 100%
D) 121%
E) 245%
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In combustion analysis, the oxygen content of a substance is equal to the total oxygen in the CO2 and H2O collected in the absorbers.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molar mass of (NH4) 3AsO4.
A) 417.80 g/mol
B) 193.03 g/mol
C) 165.02 g/mol
D) 156.96 g/mol
E) 108.96 g/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Magnesium reacts with iron(III) chloride to form magnesium chloride (which can be used in fireproofing wood and in disinfectants) and iron. 3Mg(s) + 2FeCl3(s) → 3MgCl2(s) + 2Fe(s)
A mixture of 41.0 g of magnesium ( 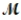 = 24.31 g/mol) and 175 g of iron(III) chloride (
= 24.31 g/mol) and 175 g of iron(III) chloride ( 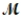 = 162.2 g/mol) is allowed to react. What mass of magnesium chloride = 95.21 g/mol) is formed?
= 162.2 g/mol) is allowed to react. What mass of magnesium chloride = 95.21 g/mol) is formed?
A) 68.5 g MgCl2
B) 77.0 g MgCl2
C) 71.4 g MgCl2
D) 107 g MgCl2
E) 154 g MgCl2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the molar mass of rubidium carbonate, Rb2CO3.
A) 340.43 g/mol
B) 255.00 g/mol
C) 230.94 g/mol
D) 145.47 g/mol
E) 113.48 g/mol
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum will react with bromine to form aluminum bromide (used as an acid catalyst in organic synthesis) . Al(s) + Br2(l) → Al2Br6(s) [unbalanced] How many moles of Al are needed to form 2.43 mol of Al2Br6?
A) 7.29 mol
B) 4.86 mol
C) 2.43 mol
D) 1.62 mol
E) 1.22 mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Constitutional (structural) isomers have the same empirical formula but different molecular formulas.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sulfur trioxide can react with atmospheric water vapor to form sulfuric acid that falls as acid rain. Calculate the mass in grams of 3.65 × 1020 molecules of SO3.
A) 6.06 × 10-4 g
B) 2.91 × 10-2 g
C) 4.85 × 10-2 g
D) 20.6 g
E) 1650 g
G) A) and D)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Terephthalic acid, used in the production of polyester fibers and films, is composed of carbon, hydrogen, and oxygen. When 0.6943 g of terephthalic acid was subjected to combustion analysis it produced 1.471 g CO2 and 0.226 g H2O. What is its empirical formula?
A) C2H3O4
B) C3H4O2
C) C4H3O2
D) C5H12O4
E) C2H2O
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Alkanes are compounds of carbon and hydrogen with the general formula CnH2n+2. An alkane component of gasoline has a molar mass of between 125 and 130 g/mol. What is the value of n for this alkane?
A) 4
B) 9
C) 10
D) 13
E) 14
G) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
What is the mass in grams of 0.250 mol of the common antacid calcium carbonate?
A) 4.00 × 102 g
B) 25.0 g
C) 17.0 g
D) 4.00 × 10-2 g
E) 2.50 × 10-3 g
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound containing chromium and silicon contains 73.52 mass percent chromium. Determine its empirical formula.
A) CrSi3
B) Cr2Si3
C) Cr3Si
D) Cr3Si2
E) Cr2S
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum sulfate, Al2(SO4) 3, is used in tanning leather, purifying water, and manufacture of antiperspirants. Calculate its molar mass.
A) 450.06 g/mol
B) 342.15 g/mol
C) 315.15 g/mol
D) 278.02 g/mol
E) 74.98 g/mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydroxylamine nitrate contains 29.17 mass % N, 4.20 mass % H, and 66.63 mass O. If its molar mass is between 94 and 98 g/mol, what is its molecular formula?
A) NH2O5
B) N2H4O4
C) N3H3O3
D) N4H8O2
E) N2H2O4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Balance the following equation: Ca3(PO4) 2(s) + SiO2(s) + C(s) → CaSiO3(s) + CO(g) + P4(s)
A) Ca3(PO4) 2(s) + 3SiO2(s) + 8C(s) → 3CaSiO3(s) + 8CO(g) + P4(s)
B) Ca3(PO4) 2(s) + 3SiO2(s) + 14C(s) → 3CaSiO3(s) + 14CO(g) + P4(s)
C) Ca3(PO4) 2(s) + 3SiO2(s) + 8C(s) → 3CaSiO3(s) + 8CO(g) + 2P4(s)
D) 2Ca3(PO4) 2(s) + 6SiO2(s) + 10C(s) → 6CaSiO3(s) + 10CO(g) + P4(s)
E) 2Ca3(PO4) 2(s) + 6SiO2(s) + 10C(s) → 6CaSiO3(s) + 10CO(g) + 4P4(s)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of hydrogen atoms in 0.050 mol of C3H8O3 is
A) 3.0 × 1022 H atoms.
B) 1.2 × 1023 H atoms.
C) 2.4 × 1023 H atoms.
D) 4.8 × 1023 H atoms.
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 70
Related Exams