A) Na[Ni(CN) 4]
B) Na[Ni(CN) 4]2
C) Na2[Ni(CN) 4]
D) Na4[Ni(CN) 4]
E) None of these choices are correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following coordination numbers applies to octahedral complexes?
A) 4
B) 5
C) 6
D) 8
E) None of these choices are correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the presence of a strong octahedral ligand field, the number of unpaired electrons in Co(III) will be
A) 0.
B) 2.
C) 4.
D) 6.
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the formation of a transition metal complex, the central metal atom or ion acts as
A) an Arrhenius acid.
B) a Bronsted-Lowry acid.
C) a Bronsted-Lowry base.
D) a Lewis acid.
E) a Lewis base.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands is most likely to form a high spin octahedral complex with cobalt(II) ?
A) CN-
B) en (ethylenediamine)
C) NO2-
D) CO
E) I?
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions is least likely to form colored compounds?
A) Mn2+
B) Cr5+
C) Sc3+
D) Fe3+
E) Co2+
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands could participate in linkage isomerism?
A) NH3
B) H2O
C) NH4+
D) NO2-
E) ethylenediamine
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following species could exist as isomers?
A) [Co(H2O) 4Cl2]+
B) [Pt(NH3) Br3]-
C) [Pt(en) Cl2]
D) [Pt(NH3) 3Cl]+
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The oxidation and coordination numbers of cobalt in the compound [Co(NH3) 5Cl]Cl2 are,respectively
A) 2 and 6.
B) 2 and 8.
C) 3 and 6.
D) 3 and 8.
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the oxidation states of chromium has the largest valence-state electronegativity?
A) chromium(0)
B) chromium(II)
C) chromium(III)
D) chromium(IV)
E) chromium(VI)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a coordination compound involving a complex ion of square planar geometry, which of the following types of isomerism is/are never possible?
A) Geometric
B) Optical
C) Linkage
D) Coordination
E) More than one of these
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures, each involving ethylene diamine and two different, unidentate ligands X and Y. 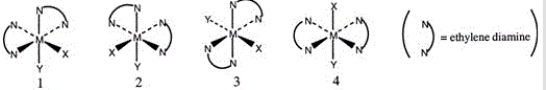 Which one of the following statements about these structures is incorrect?
Which one of the following statements about these structures is incorrect?
A) Structures 1 and 2 are optical isomers.
B) Structures 1 and 3 are optical isomers.
C) Structures 1 and 3 are different complexes.
D) Structures 1 and 4 are geometrical isomers.
E) Structures 3 and 4 are the same complex.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A characteristic of ligands is that
A) they are Lewis acids.
B) they are Lewis bases.
C) they are ions.
D) they are electron pair acceptors.
E) they are Brønsted-Lowry acids.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The maximum oxidation state of an element in the first transition series never exceeds its group number.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the spectrochemical series, which one of the following ligands has the strongest field?
A) H2O
B) CN-
C) NH3
D) OH-
E) Cl-
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ground state electronic configuration of Zn2+ is
A) [Ar]4s23d8.
B) [Ar]4s23d10.
C) [Ar]4s13d9.
D) [Ar]3d10.
E) [Ar]3d8.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aluminum reacts with oxygen in the air to form a protective oxide coating. Silver also reactswith compounds in air to form a black coating. What substance is formed?
A) silver oxide
B) silver chloride
C) silver sulfide
D) silver carbonate
E) silver nitride
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the highest possible oxidation state for molybdenum, Mo?
A) +2
B) +4
C) +6
D) +8
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A feature of transition metal chemistry is that these elements exhibit multiple oxidation states. Which one of the following elements exhibits the smallest number of different oxidation states?
A) Ti
B) Cr
C) Mn
D) Co
E) Zn
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Octahedral complexes can exhibit geometric, optical, and linkage isomerism.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 82
Related Exams