A) Co(OH) 2; [OH-] = 6.9 × 10-6 M
B) Co(OH) 2; [OH-] = 2.6 × 10-7 M
C) Cu(OH) 2; [OH-] = 1.8 × 10-7 M
D) Cu(OH) 2; [OH-] = 1.1 × 10-9 M
E) Cu(OH) 2; [OH-] = 1.0 × 10-17 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A lab technician adds 0.015 mol of KOH to 1.00 L of 0.0010 M Ca(NO3) 2. Ksp = 6.5 × 10-6 for Ca(OH) 2) . Which of the following statements is correct?
A) Calcium hydroxide precipitates until the solution is saturated.
B) The solution is unsaturated and no precipitate forms.
C) The concentration of calcium ions is reduced by the addition of the hydroxide ions.
D) One must know Ksp for calcium nitrate to make meaningful predictions on this system.
E) The presence of KOH will raise the solubility of Ca(NO3) 2.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 20.0-mL sample of 0.30 M HClO was titrated with 0.30 M NaOH. The following data were collected during the titration. mL NaOH added 5.00 10.00 1.00 2.00 PH 6.98 7.46 7.93 10.31 What is the Ka for HClO?
A) 1.1 × 10-7
B) 3.5 × 10-8
C) 1.2 × 10-8
D) 4.9 × 10-11
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a buffer that consists of 0.45 M CH3COOH and 0.35 M CH3COONa? Ka = 1.8 × 10-5
A) 4.49
B) 4.64
C) 4.85
D) 5.00
E) 5.52
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lab technician Anna Lytic adds 2.20 mol KOH to 1.00 L of 0.5 M Al(NO3) 3. What is the concentration of aluminum ions after the aluminum nitrate has reacted with the potassium hydroxide? Kf = 3.0 × 1033 for Al(OH) 4-
A) 1.8 × 10-7 M
B) 9.1 × 10-18 M
C) 1.0 × 10-31 M
D) 3.3 × 10-34 M
E) 7.1 × 10-36 M
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is the best representation of the titration curve that will be obtained in the titration of a weak acid (0.10 mol L-1) with a strong base of the same concentration? 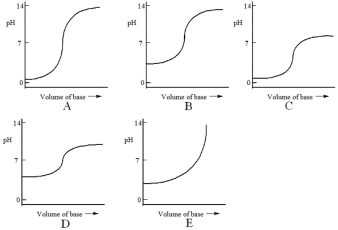
A) A
B) B
C) C
D) D
E) E
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following acids should be used to prepare a buffer with a pH of 4.5?
A) HOC6H4OCOOH, Ka = 1.0 × 10-3
B) C6H4(COOH) 2, Ka = 2.9 × 10-4
C) CH3COOH, Ka = 1.8 × 10-5
D) C5H5O5COOH, Ka = 4.0 × 10-6
E) HBrO, Ka = 2.3 × 10-9
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a buffer that consists of 0.30 M HCOOH and 0.20 M HCOONa? For HCOOH, Ka = 1.7 × 10-4
A) 1.1 × 10-4 M
B) 2.6 × 10-4 M
C) 4.3 × 10-4 M
D) 6.7 × 10-5 M
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Increasing the concentrations of the components of a buffer solution will increase the buffer capacity.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of silver chromate, Ag2CrO4, in 0.005 M Na2CrO4. Ksp = 2.6 × 10-12
A) 1.4 × 10-4 M
B) 3.4 × 10-5 M
C) 1.1 × 10-5 M
D) 1.6 × 10-6 M
E) < 1.0 × 10-6 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
For a diprotic acid H2A, the relationship Ka1 > Ka2 is always true.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following indicators would be the best to use when 0.050 M benzoic acid (Ka = 6.6 × 10-5) is titrated with 0.05 M NaOH?
A) bromphenol blue, pH range: 3.0-4.5
B) bromcresol green, pH range: 3.8-5.4
C) alizarin, pH range: 5.7-7.2
D) phenol red, pH range: 6.9-8.2
E) phenolphthalein, pH range: 8.0-10.1
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Write the ion product expression for calcium phosphate, Ca3(PO4) 2.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following aqueous mixtures would be a buffer system?
A) HCl, NaCl
B) HNO3, NaNO3
C) H3PO4, H2PO4-
D) H2SO4, CH3COOH
E) NH3, NaOH
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of barium carbonate, BaCO3, in pure water. Ksp = 2.0 × 10-9
A) 1.3 × 10-3 M
B) 3.2 × 10-5 M
C) 2.2 × 10-5 M
D) 4.5 × 10-5 M
E) 4.0 × 10-18 M
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What will be the effect of adding 0.5 mL of 0.1 M NaOH to 100 mL of an acetate buffer in which [CH3COOH] = [CH3COO-] = 0.5 M?
A) The pH will increase slightly.
B) The pH will increase significantly.
C) The pH will decrease slightly.
D) The pH will decrease significantly.
E) Since it is a buffer solution, the pH will not be affected.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the solubility of strontium fluoride, SrF2, in pure water. Ksp = 2.6 × 10-9
A) 1.4 × 10-3 M
B) 3.4 × 10-4 M
C) 8.7 × 10-4 M
D) 5.1 × 10-5 M
E) < 1.0 × 10-5 M
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of magnesium phosphate is 2.27 × 10-3 g/1.0 L of solution. What is the Ksp for Mg3(PO4) 2?
A) 6.5 × 10-12
B) 6.0 × 10-14
C) 5.2 × 10-24
D) 4.8 × 10-26
E) 1.0 × 10-26
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the [H3O+] in a solution that consists of 1.2 M HClO and 2.3 M NaClO? Ka = 3.5 × 10-8
A) 7.8 × 10-9 M
B) 1.8 × 10-8 M
C) 6.7 × 10-8 M
D) 1.6 × 10-7 M
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The solubility of aluminum hydroxide in water ______________ when dilute nitric acid is added to it.
A) increases
B) decreases
C) does not change
D) first increases, then decreases
E) first decreases, then increases
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 114
Related Exams