B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium fluoride is used for frosting glass. Calculate the molarity of a solution prepared by dissolving 78.6 g of KF in enough water to produce 225 mL of solution.
A) 0.304 M
B) 0.349 M
C) 1.35 M
D) 3.29 M
E) 6.01 M
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Human blood has a molar concentration of solutes of 0.30 M. What is the osmotic pressure of blood at 25°C?
A) 0.012 atm
B) 0.62 atm
C) 6.8 atm
D) 7.3 atm
E) >10. atm
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If a solute dissolves in an endothermic process
A) H bonds must exist between solvent and solute.
B) strong ion-dipole forces must exist in the solution.
C) the solute must be a gas.
D) the entropy of the solution is immaterial.
E) the entropy of the solution must be greater than that of its pure components.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based only on the relative lattice energies of the compounds below, which one would be expected to have the lowest solubility in water?
A) NaBr
B) CaS
C) NaOH
D) KI
E) CsCl
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Heats of solution may be either positive or negative.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Gases with high boiling points tend to be more soluble in water than ones with low boiling points.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The solubility of gases in water increases with increase in the pressure of the gas.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Benzaldehyde ( 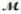 = 106.1 g/mol) , also known as oil of almonds, is used in the manufacture of dyes and perfumes and in flavorings. What would be the freezing point of a solution prepared by dissolving 75.00 g of benzaldehyde in 850.0 g of ethanol? Kf = 1.99°C/m, freezing point of pure ethanol = -117.3°C.
= 106.1 g/mol) , also known as oil of almonds, is used in the manufacture of dyes and perfumes and in flavorings. What would be the freezing point of a solution prepared by dissolving 75.00 g of benzaldehyde in 850.0 g of ethanol? Kf = 1.99°C/m, freezing point of pure ethanol = -117.3°C.
A) -117.5°C
B) -118.7°C
C) -119.0°C
D) -120.6°C
E) < -121°C
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Raoult's Law relates the vapor pressure of a solvent above a solution to the mole fraction of the solvent and the vapor pressure of the pure solvent.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Isoamyl salicylate ( 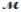 = 208.25 g/mol) has a pleasant aroma and is used in perfumes and soaps. Which of the following combinations gives a 0.75 m solution of isoamyl salicylate in ethyl alcohol (d = 0.7893 g/mL) ?
= 208.25 g/mol) has a pleasant aroma and is used in perfumes and soaps. Which of the following combinations gives a 0.75 m solution of isoamyl salicylate in ethyl alcohol (d = 0.7893 g/mL) ?
A) 117.2 g isoamyl salicylate in 950.0 mL of ethyl alcohol
B) 117.2 g isoamyl salicylate in 750.0 mL of ethyl alcohol
C) 117.2 g isoamyl salicylate in 750.0 mL of solution
D) 117.2 g isoamyl salicylate in 592.0 g of ethyl alcohol
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the weakest electrolyte from the following set.
A) Na2 SO4
B) KCl
C) CH3 CH2 COOH, propionic acid
D) CaCl2
E) LiOH
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs of ions is arranged so that the ion with the larger (i.e., more negative) heat of hydration is listed first?
A) Br-, K+
B) Mg2+, Sr 2+
C) Ca2+, Sc3+
D) Na+, Li+
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Colligative properties depend on
A) the chemical properties of the solute.
B) the chemical properties of the solvent.
C) the masses of the individual ions.
D) the molar mass of the solute.
E) the number of particles dissolved.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution of ethanol (C2H6O) in water is sometimes used as a disinfectant. 1.00 L of this solution contains 553 g of ethanol and 335 g of water. What is the molality of the ethanol in this solution?
A) 0.392 m
B) 0.608 m
C) 13.5 m
D) 33.6 m
E) 35.8 m
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution of sucrose (sugar) in water is in equilibrium with solid sucrose. If more solid sucrose is now added, with stirring,
A) the concentration of the solution will increase.
B) the concentration of the solution will decrease.
C) the concentration of the solution will remain the same.
D) the volume of solution will increase.
E) a supersaturated solution will be produced.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diethyl ether has a vapor pressure of 400.0 torr at 18°C. When a sample of benzoic acid is dissolved in ether, the vapor pressure of the solution is 342 torr. What is the mole fraction of benzoic acid in the solution?
A) 0.0169
B) 0.0197
C) 0.145
D) 0.855
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
From the following list of aqueous solutions and water, select the one with the highest boiling point.
A) 1.0 m KNO3
B) 0.75 m NaCl
C) 0.75 m CuCl2
D) 2.0 m C12H22O11 (sucrose)
E) pure water
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Children under the age of 6 with more than 0.10 ppm of lead in their blood can suffer a reduction in I.Q. or have behavior problems. What is the molality of a solution which contains 0.10 ppm of lead?
A) 4.8 × 10-10 m
B) 4.8 × 10-7 m
C) 4.8 × 10-4 m
D) 4.8 × 10-1 m
E) None of these choices are correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the vapor pressure of a solution prepared by dissolving 0.500 mol of a non-volatile solute in 275 g of hexane ( 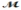 = 86.18 g/mol) at 49.6°C. P°hexane = 400.0 torr at 49.6°C.
= 86.18 g/mol) at 49.6°C. P°hexane = 400.0 torr at 49.6°C.
A) 54 torr
B) 154 torr
C) 246 torr
D) 346 torr
E) 400. torr
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 96
Related Exams