A) bent
B) linear
C) trigonal planar
D) T-shaped
E) trigonal pyramidal
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules has a zero dipole moment?
A) SO2
B) HCl
C) CS2
D) CO
E) Cl2O
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In a Lewis structure for a molecule or ion, the sum of the formal charges on the atoms is equal to the charge on the molecule or ion.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Considering all the bonds in a molecule with trigonal bipyramidal geometry, what are the bond angles present?
A) 120° only
B) 90° only
C) 180° only
D) 60° and 90° only
E) 90°, 120°, and 180°
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules contains a double bond?
A) N2
B) PCl5
C) CH2O
D) C2H2
E) I2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct Lewis structure for NOCl, a reactive material used as an ionizing solvent.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct Lewis structure for nitrogen trifluoride, NF3.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of BeH2 as predicted by the VSEPR theory?
A) linear
B) bent
C) angular
D) trigonal
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many faces and how many vertexes (corners) , respectively, are there in a trigonal bipyramid?
A) 4 and 4
B) 5 and 5
C) 5 and 6
D) 6 and 5
E) 6 and 8
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In formaldehyde, CH2O, both the formal charge and the oxidation number of carbon are zero.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular symmetry around the carbons in CCl2CH2 as predicted by the VSEPR theory?
A) linear
B) trigonal planar
C) V-shaped
D) tetrahedral
E) trigonal pyramidal
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles around nitrogen in N2F2 using the molecular shape given by the VSEPR theory. (The two N atoms are the central atoms.)
A) 90°
B) 109°
C) 120°
D) 180°
E) between 120 and 180°
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX6 will have a ______ molecular shape.
A) tetrahedral
B) trigonal planar
C) trigonal bipyramidal
D) hexagonal
E) octahedral
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules and ions will have a planar geometry?
A) PCl3
B) BF4-
C) XeF4
D) BrF5
E) H3O+
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formal charge on Cl in the structure shown for the perchlorate ion is 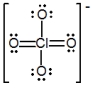
A) -2
B) -1
C) 0
D) +1
E) +2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX2E2 will have a _____ molecular shape.
A) linear
B) bent
C) trigonal planar
D) tetrahedral
E) see-saw
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Oxygen difluoride is a powerful oxidizing and fluorinating agent. Select its Lewis structure.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices are correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following is the best Lewis structure a resonance structure?
A) SO3
B) BF3
C) I3-
D) SCO (C = central atom)
E) SO32-
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The molecule AX2, where A and X are different elements, will have a dipole moment if the molecule is bent.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 98
Related Exams