A) 16.2 g
B) 33.4 g
C) 40.6 g
D) 81.3 g
E) 163 g
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following balanced redox reaction Mn2+(aq) + S2O82¯(aq) + 2H2O(l) MnO2(s) + 4H+(aq) + 2SO42¯(aq) Which of the following statements is true?
A) Mn2+(aq) is the oxidizing agent and is reduced.
B) Mn2+(aq) is the oxidizing agent and is oxidized.
C) Mn2+(aq) is the reducing agent and is oxidized.
D) Mn2+(aq) is the reducing agent and is reduced.
E) Manganese does not change its oxidation number in this reaction.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two cells are connected in series, so that the same current flows through two electrodes where the following half-reactions occur Cu2+(aq) + 2e¯ Cu(s) and Ag+(aq) + e¯ Ag(s) For every 1.00 g of copper produced in the first process, how many grams of silver will be produced in the second one?
A) 0.294 g
B) 0.588 g
C) 0.850 g
D) 1.70 g
E) 3.40 g
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A voltaic cell consists of a Hg/Hg22+ electrode (E = 0.85 V) and a Sn/Sn2+ electrode (E = -0.14 V) . Calculate [Sn2+] if [Hg22+] = 0.24 M and Ecell = 1.04 V at 25 C.
A) 0.0001 M
B) 0.0007 M
C) 0.005 M
D) 0.03 M
E) 0.05 M
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In a fuel cell, an external source of electrical power is used to drive a non-spontaneous reaction in which a fuel is produced.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the value of the equilibrium constant for the cell reaction below at 25 C?
E cell = 0.61 V 2Cr(s) + 3Pb2+(aq) 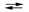 3Pb(s) + 2Cr3+(aq)
3Pb(s) + 2Cr3+(aq)
A) 4.1 * 1020
B) 8.2 * 1030
C) 3.3* 1051
D) 7.4 * 1061
E) > 9.9 *1099
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following half-reactions and select the strongest oxidizing agent among the substances. ![Examine the following half-reactions and select the strongest oxidizing agent among the substances. A) [PtCl<sub>4</sub>]<sup>2</sup>¯(aq) B) RuO<sub>4</sub>(s) C) HFeO<sub>4</sub>¯(aq) D) H<sub>4</sub>XeO<sub>6</sub>(aq) E) Cl¯(aq)](https://d2lvgg3v3hfg70.cloudfront.net/TB7799/11eb16b2_ffae_3abd_984d_27bba5c6e3e0_TB7799_00.jpg)
A) [PtCl4]2¯(aq)
B) RuO4(s)
C) HFeO4¯(aq)
D) H4XeO6(aq)
E) Cl¯(aq)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by dissolving 32.0 g of NiSO4 in water. What current would be needed to deposit all of the nickel in 5.0 hours?
A) 1.1 A
B) 2.2 A
C) 3.3 A
D) 4.4 A
E) 5.5 A
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following balanced redox reaction 3CuO(s) + 2NH3(aq) N2(g) + 3H2O(l) + 3Cu(s) Which of the following statements is true?
A) CuO(s) is the oxidizing agent and copper is reduced.
B) CuO(s) is the oxidizing agent and copper is oxidized.
C) CuO(s) is the reducing agent and copper is oxidized.
D) CuO(s) is the reducing agent and copper is reduced.
E) CuO(s) is the oxidizing agent and N2(g) is the reducing agent.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following half-reactions and select the strongest reducing agent among the species listed. 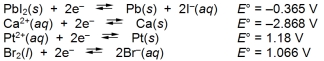
A) Pb(s)
B) Ca(s)
C) Pt(s)
D) Br¯(aq)
E) Pt2+(aq)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A battery that cannot be recharged is a
A) fuel cell.
B) primary battery.
C) secondary battery.
D) simple battery.
E) flow battery.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Examine the following half-reactions and select the weakest reducing agent among the substances. 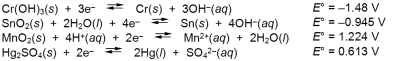
A) Cr(s)
B) Sn(s)
C) Mn2+(aq)
D) Hg(l)
E) OH¯(aq)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the E cell for the cell represented by the combination of the following half-reactions? 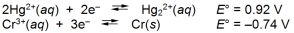
A) -0.18 V
B) 0.18 V
C) 1.28 V
D) 1.66 V
E) 2.12 V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate E cell for the reaction of nickel(II) ions with cadmium metal at 25 C. K = 1.17 *105. Ni2+(aq) + Cd(s) Cd2+(aq) + Ni(s)
A) 0.075 V
B) 0.10 V
C) 0.12 V
D) 0.15 V
E) 0.30 V
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The redox reaction of peroxydisulfate with iodide has been used for many years as part of the iodine clock reaction which introduces students to kinetics. If E cell = 1.587 V and E of the cathode half-cell is 0.536 V, what is E of the anode half-cell? 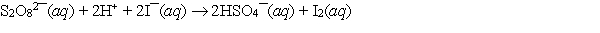
A) -1.051 V
B) -2.123 V
C) 1.051 V
D) 2.123 V
E) None of these choices is correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of silver will be formed when 15.0 A are passed through molten AgCl for 25.0 minutes?
A) 0.419 g
B) 6.29 g
C) 12.6 g
D) 25.2 g
E) 33.4 g
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the reaction of iodine with manganese dioxide:
3I2(s) + 2MnO2(s) + 8OH¯(aq) 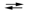 6I¯(aq) + 2MnO4¯(aq) + 4H2O(l)
The equilibrium constant for the overall reaction is 8.30* 10¯7. Calculate G for the reaction at 25 C.
6I¯(aq) + 2MnO4¯(aq) + 4H2O(l)
The equilibrium constant for the overall reaction is 8.30* 10¯7. Calculate G for the reaction at 25 C.
A) -15.1 kJ
B) -34.7 kJ
C) 15.1 kJ
D) 34.7 kJ
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the non-aqueous cell reaction  for which E cell = 2.35 V at 200 C. G at this temperature is
for which E cell = 2.35 V at 200 C. G at this temperature is
A) 453 kJ.
B) -453 kJ.
C) 907 kJ.
D) -907 kJ.
E) None of these choices is correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced with smallest whole number coefficients, the coefficient for the iodide ion will be _____. 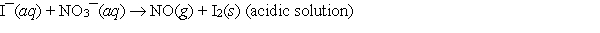
A) 2
B) 3
C) 6
D) 8
E) None of these choices is correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A concentration cell consists of two Al/Al3+ electrodes. The electrolyte in compartment A is 0.050 M Al(NO3) 3 and in compartment B is 1.25 M Al(NO3) 3. What is the voltage of the cell at 25 C?
A) 0.083 V
B) 0.062 V
C) 0.041V
D) 0.028 V
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 102
Related Exams