A) The nucleophile is too basic.
B) Reforming the carbonyl is energetically favorable.
C) The leaving group is unstable and wants to be negatively charged.
D) There is no tetrahedral intermediate.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Will the following reaction occur?
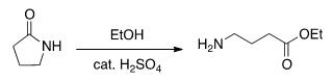
A) Yes
B) No
D) undefined
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What reagent would you use for the following transformation?

A) LiAlH4
B) DIBAL-H
C) NaBH4
D) H2, Pd/C
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Where do the carbonyl signals appear in the 13C NMR spectrum of carboxylic acid derivatives?
A) 1700 cm-1
B) 180-160 ppm
C) 2.5-3.0 ppm
D) 100-80 ppm
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How can you convert a carboxylic acid into an acid chloride?
A) Heat with hydrochloric acid.
B) React with thionyl chloride (SOCl2) .
C) React with sodium chloride.
D) React with Cl2 and FeCl3.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the direct product of the base-promoted hydrolysis of an ester?
A) A nitrile
B) A carboxylic acid
C) An amide
D) A carboxylic acid salt
Gradable: automatic
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 46 of 46
Related Exams