A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Most foodstuffs contain natural, radioactive isotopes.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is a subatomic particle closely related to the positron?
A) Proton
B) Electron
C) Negatron
D) Neutron
E) Neutrino
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Detection of radiation by a Geiger-Müller counter depends on
A) the emission of a photon from an excited atom.
B) the ability of an ionized gas to carry an electrical current.
C) the emission of a photon of light by the radioactive particle.
D) the ability of a photomultiplier tube to amplify the electrical signal from a phosphor.
E) the detection of the sound made by decay particles.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the nuclide that completes the following nuclear reaction. 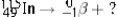
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An isotope with a high value of N/Z will tend to decay through
A) α decay.
B) β decay.
C) positron decay.
D) electron capture.
E) γ decay.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following isotopes is definitely unstable?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the missing species in the following nuclear transmutation. 
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotopes 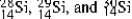 are all stable, while
are all stable, while 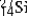 is radioactive. The mode of decay for
is radioactive. The mode of decay for 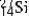 is most likely to be
is most likely to be
A) positron decay.
B) alpha decay.
C) beta decay.
D) gamma decay.
E) fission.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the missing species in the following nuclear transmutation. 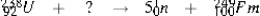
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
After 4 half-lives, the fraction of a radioactive isotope which still remains is approximately one-eighth.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The isotopes of promethium, 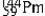 and
and 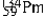 are unstable, and lie on opposite sides of the "line of stability". Which of the following combinations is most likely to represent the type of decay for these isotopes?
are unstable, and lie on opposite sides of the "line of stability". Which of the following combinations is most likely to represent the type of decay for these isotopes?
A) Promethium-144, β decay; Promethium-134, positron decay
B) Promethium-144, positron decay; Promethium-134, β decay
C) Promethium-144, positron decay; Promethium-134, electron capture
D) Promethium-144, electron capture; Promethium-134, positron decay
E) Promethium-144, β decay; Promethium-134, γ decay
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following types of radioactive decay does not produce new element?
A) Gamma emission
B) Electron capture
C) Beta emission
D) Alpha emission
E) Double beta emission
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
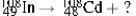 In the equation above, what particle or type of radiation needs to be included on the right-hand side in order to balance it?
In the equation above, what particle or type of radiation needs to be included on the right-hand side in order to balance it?
A) Alpha
B) Beta
C) Gamma
D) Positron
E) Proton
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The r-process occurs during supernova explosions.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the nuclide that completes the following nuclear reaction. 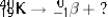
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
No alpha decay is observed for isotopes of elements with Z < 83.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium-21 will emit positrons each having an energy of 4.0 × 10-13 J. What is this energy in MeV?
A) 4.0 × 10 -7 MeV
B) 2.5 MeV
C) 40 MeV
D) 2.5 × 10 6 MeV
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A N-14 nucleus is hit by a particle, forming a C-14 nucleus and a proton as the only products. Identify the type of particle which struck the N-14 nucleus.
A) Alpha
B) Proton
C) Electron
D) Neutron
E) Deuterium
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Who discovered radioactivity?
A) Geiger
B) Curie
C) Roentgen
D) Becquerel
E) Rutherford
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 82
Related Exams