A) It is non-spontaneous at all T.
B) It is spontaneous at high T.
C) It is spontaneous at low T.
D) It is spontaneous at all T.
E) None of these choices are correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? 2NH3(g) + 2ClF3(g) → 6HF(g) + N2(g) + Cl2(g)
A) Δ S° ≈ 0
B) Δ S° < 0
C) Δ S° > 0
D) Δ S° = Δ H°/ T
E) More information is needed to make a reasonable prediction.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔS° for the reaction SiCl4(g) + 2Mg(s) → 2MgCl2(s) + Si(s)

A) −254.96 J/K
B) −198.02 J/K
C) 198.02 J/K
D) 254.96 J/K
E) 471.86 J/K
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔG° for the combustion of propane. C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g)
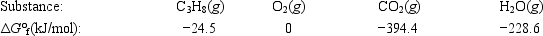
A) −2073.1 kJ
B) −1387.3 kJ
C) −598.5 kJ
D) 598.5 kJ
E) 2073.1 kJ
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔS° for the combustion of propane. C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g)
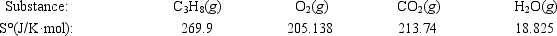
A) −100.9 J/K
B) −72.5 J/K
C) 72.5 J/K
D) 100.9 J/K
E) 877.5 J/K
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following results in a decrease in the entropy of the system?
A) O 2( g) , 300 K → O 2( g) , 400 K
B) H 2O( s) , 0°C → H 2O( l) , 0°C
C) N 2( g) , 25°C → N 2( aq) , 25°C
D) NH 3( l) , −34.5°C → NH 3( g) , −34.5°C
E) 2H 2O 2( g) → 2H 2O( g) + O 2( g)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which, if any, of the following processes is spontaneous under the specified conditions?
A) H 2O( l) → H 2O( s) at 25°C
B) CO 2( s) → CO 2( g) at 0°C
C) 2H 2O( g) → 2H 2(g) + O 2( g)
D) C(graphite) → C(diamond) at 25°C and 1 atm pressure
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You are given pure samples of ethane, C2H6(g) , and toluene, C7H8(l) . What prediction would you make concerning their standard molar entropies at 298 K?
A) S° ethane > S° toluene
B) S° ethane < S° toluene
C) S° ethane ≈ ( S° toluene) ÷ 3
D) S° ethane ≈ S° toluene
E) Since toluene is much more complex than ethane, but ethane is in the gas phase while toluene is a liquid, any of these predictions can be confidently made without further information or calculations.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? C2H5OH(l) + 3O2(g) → 2CO2(g) + 3H2O(l)
A) Δ S° ≈ 0
B) Δ S° < 0
C) Δ S° > 0
D) Δ S° = Δ H°/ T
E) More information is needed to make a reasonable prediction.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is necessary for a process to be spontaneous?
A) Δ H sys < 0
B) Δ S sys > 0
C) Δ S surr < 0
D) Δ S univ > 0
E) Δ G sys = 0
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔS° for the reaction 4Cr(s) + 3O2(g) → 2Cr2O3(s)
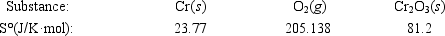
A) −548.1 J/K
B) −147.7 J/K
C) 147.7 J/K
D) 310.1 J/K
E) 548.1 J/K
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? KCl(s) → K+(aq) + Cl−(aq)
A) Δ S° ≈ 0
B) Δ S° < 0
C) Δ S° > 0
D) Δ S° = Δ H°/ T
E) More information is needed to make a reasonable prediction.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the figure that shows ΔG° for a chemical process plotted against absolute temperature. 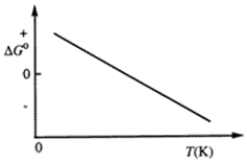 Which one of the following is an incorrect conclusion, based on the information in the diagram?
Which one of the following is an incorrect conclusion, based on the information in the diagram?
A) Δ H° > 0
B) Δ S° > 0
C) The reaction is spontaneous at high temperatures.
D) Δ S° increases with temperature while Δ H° remains constant.
E) There exists a certain temperature at which Δ H° = TΔ S°.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The second law of thermodynamics tells us that
A) the entropy of the universe is constant.
B) entropy is neither created nor destroyed.
C) the universe proceeds toward a state of lower entropy.
D) the universe proceeds toward a state of higher entropy.
E) the universe cannot create entropy.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the figure that shows ΔG° for a chemical process plotted against absolute temperature. From this plot, it is reasonable to conclude that 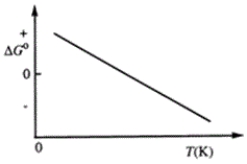
A) Δ H° > 0, Δ S° > 0
B) Δ H° > 0, Δ S° < 0
C) Δ H° < 0, Δ S° > 0
D) Δ H° < 0, Δ S° < 0
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The temperature at which the following process reaches equilibrium at 1.0 atm is the normal melting point for phosphoric acid. H3PO4(s) ⇄ H3PO4(l)
Use the following thermodynamic information at 298 K to determine this temperature.
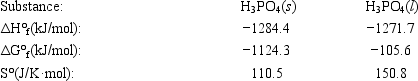
A) 286 K
B) 305 K
C) 315 K
D) 347 K
E) 3170 K
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formation constant for the reaction Ag+(aq) + 2NH3(aq) ⇄ Ag(NH3) 2+(aq) Is Kf = 1.7 × 107 at 25°C. What is ΔG° at this temperature?
A) −1.5 kJ
B) −3.5 kJ
C) −18 kJ
D) −23 kJ
E) −41 kJ
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true for a system at equilibrium?
A) Δ S° sys = Δ S° surr
B) Δ S° sys = −Δ S° surr
C) Δ S° sys = Δ S° surr = 0
D) Δ S° univ > 0
E) None of these choices are correct.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a chemical reaction to be spontaneous only at high temperatures, which of the following conditions must be met?
A) Δ S° > 0, Δ H° > 0
B) Δ S° > 0, Δ H° < 0
C) Δ S° < 0, Δ H° < 0
D) Δ S° < 0, Δ H° > 0
E) Δ G° > 0
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following conditions will ensure that a chemical reaction will definitely proceed in the forward direction, toward products?
A) Δ H > 0
B) Δ H < 0
C) Δ S sys > 0
D) Δ S surr > Δ S sys
E) Δ S > Δ H/ T
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 85
Related Exams