A) Surface tension
B) Capillary action
C) Viscosity
D) Adhesion
E) Cohesion
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has a boiling point that does not fit the general trend?
A) NH 3
B) PH 3
C) AsH 3
D) SbH 3
E) BiH 3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metal such with a face-centered cubic lattice will have __________ atom(s) per unit cell.
A) 1
B) 2
C) 3
D) 4
E) 10
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Comparing the energies of the following intermolecular forces on a kJ/mol basis, which would normally have the highest energy (i.e., be the strongest force) ?
A) Ion-induced dipole
B) Dipole-induced dipole
C) Ion-dipole
D) Dipole-dipole
E) Dispersion
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs of substances is arranged so that the one with higher viscosity is listed first? 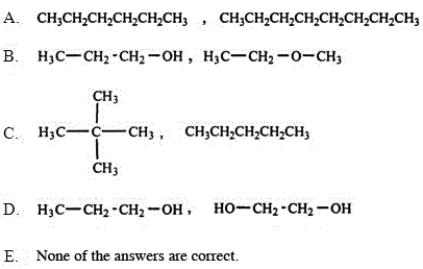
A) A
B) B
C) C
D) D
E) E
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances will have hydrogen bonds between molecules?
A) (CH 3) 3N
B) CH 3-O-CH 3
C) CH 3CH 2-OH
D) CH 3CH 2-F
E) HI
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true about kinetic energy, Ek, and potential energy, Ep, when ethyl alcohol at 40°C is compared with ethyl alcohol at 20°C?
A) E k(40°C) < E k(20°C) ; E p(40°C) ≈ E p(20°C)
B) E k(40°C) > E k(20°C) ; E p(40°C) ≈ E p(20°C)
C) E p(40°C) < E p(20°C) ; E k(40°C) ≈ E k(20°C)
D) E p(40°C) > E p(20°C) ; E k(40°C) ≈ E k(20°C)
E) E p(40°C) > E p(20°C) ; E k(40°C) > E k(20°C)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A temperature increase causes __________ in the conductivity of a conductor.
A) a decrease
B) an increase
C) an increase or decrease (depending on the conductor)
D) a modulation
E) no change
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following should have the highest surface tension at a given temperature?
A) CH 4
B) CF 4
C) CCl 4
D) CBr 4
E) CI 4
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Iron crystallizes in the body-centered cubic lattice. What is the coordination number for Fe?
A) 4
B) 6
C) 8
D) 10
E) 12
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of octane in equilibrium with its vapor in a closed 1.0-L container has a vapor pressure of 50.0 torr at 45°C. The container's volume is increased to 2.0 L at constant temperature and the liquid/vapor equilibrium is reestablished. What is the vapor pressure?
A) > 50.0 torr
B) 50.0 torr
C) 25.0 torr
D) The mass of the octane vapor is needed to calculate the vapor pressure.
E) The external pressure is needed to calculate the vapor pressure.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The maximum number of phases of a single substance which can coexist in equilibrium is two.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Neon atoms are attracted to each other by
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonding.
E) intramolecular forces.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The surface tension of water is lowered when a detergent is present in solution.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane is a component of fuel used in internal combustion engines. The dominant intermolecular forces in octane are
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonds.
E) carbon-hydrogen bonds.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Showing 81 - 95 of 95
Related Exams