B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the best Lewis structure for ClCN.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX4E2 will have a __________ molecular shape.
A) tetrahedral
B) square pyramidal
C) square planar
D) octahedral
E) see-saw
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of BCl3 as predicted by the VSEPR theory?
A) Linear
B) Trigonal planar
C) Bent
D) Tetrahedral
E) Trigonal pyramidal
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX6 will have a __________ molecular shape.
A) tetrahedral
B) trigonal planar
C) trigonal bipyramidal
D) hexagonal
E) octahedral
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of NO2− as predicted by the VSEPR theory?
A) Linear
B) Trigonal planar
C) Bent
D) Tetrahedral
E) Resonant
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electron pairs are shared between the carbon atoms in C2H4?
A) 5
B) 4
C) 3
D) 2
E) 1
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX2E will have a __________ molecular shape.
A) bent
B) see-saw
C) trigonal planar
D) T-shaped
E) trigonal pyramidal
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles around carbon in C2I2 using the molecular shape given by the VSEPR theory.
A) 90°
B) 109°
C) 120°
D) 180°
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to decide which one of the following ions and molecules is likely to be planar. (The central atom is always first in the formula.)
A) BrF 3
B) H 3O +
C) PCl 3
D) SO 4 2−
E) SF 4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following does the nitrogen atom have a formal charge of −1?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles around nitrogen in N2F2 using the molecular shape given by the VSEPR theory. (The two N atoms are the central atoms.)
A) 90°
B) 109°
C) 120°
D) 180°
E) between 120 and 180°
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of ClCN as predicted by the VSEPR theory? (Carbon is the central atom.)
A) Linear
B) Bent
C) Angular
D) Trigonal
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules and ions will have a planar geometry?
A) PCl 3
B) BF 4 −
C) XeF 4
D) BrF 5
E) H 3O +
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of the thiocyanate anion, SCN−, as predicted by the VSEPR theory? (Carbon is the central atom.)
A) Linear
B) Bent
C) Angular
D) Trigonal
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of BeH2 as predicted by the VSEPR theory?
A) Linear
B) Bent
C) Angular
D) Trigonal
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect? 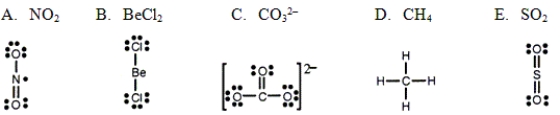
A) A
B) B
C) C
D) D
E) E
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the actual bond angles in SF3+ using the VSEPR theory.
A) more than 120°
B) exactly 120°
C) between 109° and 120°
D) between 90° and 109°
E) less than 90°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of SCl3F as predicted by the VSEPR theory?
A) Linear
B) Bent
C) See-saw
D) T-shaped
E) Trigonal pyramidal
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to decide which one of the following molecules and ions will definitely have at least one 90° bond angle in it. (In each case except water, the central atom is the first one in the formula.)
A) AlCl 4 −
B) NH 3
C) PCl 5
D) CO 2
E) H 2O
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 98
Related Exams