A) CH3NH3+
B) BF3
C) CH3O-
D) CH3OCH3
E) C and D
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What form of glutamic acid (below) predominates in a solution of
PH = 7.2?
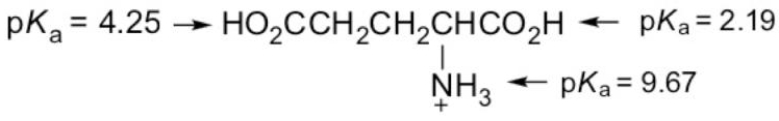
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following acid-base reactions favor the products?
A) HF + H2O ? F- + H3O+
B) ![]()
C) CH3CH3 + H2C=CH- ? CH3CH2- + H2C=CH2
D) CH3CO2H + CH3CH2O- ? CH3CO2- + CH3CH2OH
E) CH3OH + CH3CO2H ? CH3OH2+ + CH3CO2-
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following equilibrium:
CH3C  CH + B ? CH3C
CH + B ? CH3C  C- + BH+
Which of the following bases would favor products in this reaction?
C- + BH+
Which of the following bases would favor products in this reaction?
A) OH-
B) CH3O-
C) NH3
D) NH2-
E) CH3CO2-
The weakest conjugate acid produces the strongest base (ammonia has a pKa of 36) .
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a Brønsted acid?
A) AlCl3
B) CH3OH
C) HNO3
D) BF3
E) A and D
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the most acidic? Approximate pKa
A) CH3OH
B) CH3OH2+
C) CH3NH2
D) CH3NH3+
E) CH3CO2H
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a factor in determining the acidity of a molecule?
A) electronegativity
B) size
C) hybridization
D) electron delocalization
E) all of the above
G) A) and C)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
Which of the following is the strongest acid?
A) CH3CO2H
B) BrCH2CO2H
C) ClCH2CH2CO2H
D) ClCH2CH2CH2CO2H
E) CICH2CO2H
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a Lewis acid?
A) AlCl3
B) CH3OH
C) HNO3
D) BF3
E) all of the above
G) None of the above
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
Which compound is the conjugate base of CH3OH?
A) CH3OH2+
B) CH3O-
C) -CH2OH
D) HO-
E) H2O
G) A) and B)
Correct Answer

verified
B
Correct Answer
verified
Showing 1 - 10 of 10
Related Exams