A) A2 + 2 B → A2B2
B) 8 A + 4 B2 → 4 A2B2
C) 2 A + B2 → A2B2
D) 4 A + 4 B2 → 4 A2B2
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The empirical formula of a compound that contains 82.66% carbon and 17.34% hydrogen is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass of 9.00 × 1022 molecules of NH3?
A) 0.00878 g
B) 0.393 g
C) 2.55 g
D) 114 g
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following contains 52% carbon by mass?
A) C2H2
B) CH4
C) CH3OCH3
D) CO2
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Dinitrogen monoxide gas decomposes to form nitrogen gas and oxygen gas.How many grams of oxygen are formed when 20.0 g of dinitrogen monoxide decomposes?
A) 0.138 g
B) 7.27 g
C) 14.5 g
D) 29.1 g
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following diagrams represent the reaction of A2 (shaded spheres) with B2 (unshaded spheres) .How many moles of product can be made from 1.0 mol of A2 and 1.0 mol of B2? 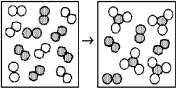
A) 0.67 mol product
B) 1.0 mol product
C) 2.0 mol product
D) 3.0 mol product
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
The number of grams in 0.250mol of urea, (NH2)2CO,is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the stoichiometric coefficient for water when the following equation is balanced using the lowest whole-number coefficients? ________ C3H8O(l) + ________ O2(g) → ________ CO2(g) + ________ H2O(l)
A) 3
B) 7
C) 6
D) 8
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Analysis of a 1.000-g sample of the oral hypoglycemic agent metformintm yielded 0.3720 g of carbon,0.0858 g of hydrogen,and 0.5422 g of nitrogen.Metformintm has a molar mass of 129.16 g/mol.What is the molecular formula of Metformintm?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the percent yield for the following reaction is 60.0%,and 45.0 g of NO2 are consumed in the reaction,how many grams of nitric acid,HNO3(aq) ,are produced? 3 NO2(g) + H2O(l) → 2 HNO3(aq) + NO(g)
A) 24.6 g
B) 41.1 g
C) 54.8 g
D) 69.3 g
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Combustion analysis of a 0.675 g sample of an unknown compound that contains only carbon,hydrogen,and oxygen gives 0.627 g of CO2 and 1.534 g of H2O.The molecular mass of the unknown is
A) C3H6O.
B) C6H12O2.
C) C9H18O3.
D) unable to be determined from this data.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has the smallest mass?
A) 6.02 × 1023 molecules of I2
B) 70.0 g of Cl2
C) 2.00 mol of F2
D) 0.040 kg of Br2
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many chloride ions are there in 3.00 mol of aluminum chloride?
A) 3.00 chloride ions
B) 9.00 chloride ions
C) 1.81 × 1024 chloride ions
D) 5.42 × 1024 chloride ions
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many cations are there in 50.0 g of sodium phosphate?
A) 1.84 × 1023 cations
B) 5.51 × 1023 cations
C) 1.97 × 1024 cations
D) 5.92 × 1024 cations
F) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
When the reaction C4H10 + O2 → CO2 + H2O is balanced using the smallest whole number coefficients,the coefficient in front of O2 is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
![-Acetone has the formula C<sub>3</sub>H<sub>6</sub>O.Which ball and stick model shown above represents acetone? [gray spheres = C,black spheres = O,unshaded spheres = H] A) model a) B) model b) C) model c) D) model d)](https://d2lvgg3v3hfg70.cloudfront.net/TB4940/11ea7e2d_d09e_aff7_a2f7_11edcd81f00c_TB4940_00_TB4940_00_TB4940_00_TB4940_00.jpg) -Acetone has the formula C3H6O.Which ball and stick model shown above represents acetone? [gray spheres = C,black spheres = O,unshaded spheres = H]
-Acetone has the formula C3H6O.Which ball and stick model shown above represents acetone? [gray spheres = C,black spheres = O,unshaded spheres = H]
A) model a)
B) model b)
C) model c)
D) model d)
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the stoichiometric coefficient for oxygen when the following equation is balanced using the lowest whole-number coefficients? ________ C3H6O2(l) + ________ O2(g) → ________ CO2(g) + ________ H2O(l)
A) 1
B) 3
C) 5
D) 7
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If unshaded spheres represent nitrogen atoms and shaded spheres represent oxygen atoms,which box represents reactants and which represents products for the reaction 2 NO2(g) → 2 NO(g) + O2(g) ? 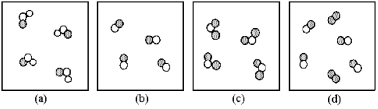
A) box (a) reactants and box (b) products
B) box (a) reactants and box (d) products
C) box (c) reactants and box (b) products
D) box (c) reactants and box (d) products
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many Fe(II) ions are there in 5.00 g of FeSO4?
A) 5.46 × 10-26 iron(II) ions
B) 1.98 × 1022 iron(II) ions
C) 1.83 × 1025 iron(II) ions
D) 4.58 × 1026 iron(II) ions
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If unshaded spheres represent nitrogen atoms and shaded spheres represent oxygen atoms,which box represents reactants and which represents products for the reaction 2 N2O(g) → 2 N2(g) + O2(g) ? 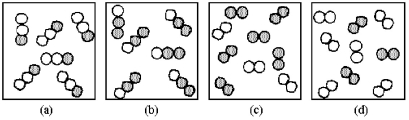
A) box (a) reactants and box (c) products
B) box (a) reactants and box (d) products
C) box (b) reactants and box (c) products
D) box (b) reactants and box (d) products
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 159
Related Exams