A) Ne
B) Rb
C) P
D) I
E) Cl
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The electrostatic energy of two charged particles is inversely proportional to the distance between them.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
When an atom is represented in a Lewis electron dot symbol,the element symbol represents ______________ and the dots represent ______________.
Correct Answer

verified
nucleus an...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
When one mole of each of the following liquids is burned,which will produce the most heat energy?
A) C6H14
B) C5H12
C) C6H14O
D) C6H12O
E) C6H10O3
G) A) and C)
Correct Answer

verified
A
Correct Answer
verified
Essay
In not more than three sentences,describe the electron arrangement responsible for bonding in Cl2 molecules.
Correct Answer

verified
This is covalent bonding.The chlorine at...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
Bond energy increases as bond order increases,for bonding between a given pair of atoms.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Electronegativity is a measure of
A) the energy needed to remove an electron from an atom.
B) the energy released when an electron is added to an atom.
C) the magnitude of the negative charge on an electron.
D) the attraction by an atom for electrons in a chemical bond.
E) the magnitude of the negative charge on a molecule.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The lattice energy of large ions is greater in magnitude than that of small ions of the same charge.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the least electronegative?
A) Si
B) Se
C) S
D) Sc
E) Sr
G) A) and C)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
In which of these substances are the atoms held together by metallic bonding?
A) CO2
B) Si
C) Br2
D) S8
E) Cr
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lattice energy of CaF2 is the energy change for which one,if any,of the following processes?
A) Ca2+(s) + 2F-(g) CaF2(g)
B) CaF2(g) CaF2(s)
C) Ca(g) + 2F(g) CaF2(s)
D) CaF2(aq) CaF2(s)
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the most electronegative?
A) S
B) Ru
C) Si
D) Te
E) Cs
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange aluminum,nitrogen,phosphorus and indium in order of increasing electronegativity.
A) Al < In < N < P
B) Al < In < P < N
C) In < Al < P < N
D) In < P < Al < N
E) None of these orders is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange calcium,rubidium,sulfur,and arsenic in order of decreasing electronegativity.
A) S > As > Rb > Ca
B) S > As > Ca > Rb
C) As > S > Rb > Ca
D) As > S > Ca > Rb
E) None of these orders is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Using appropriate,real examples to illustrate your answer,describe the correlation between bond energy and bond length for a series of varying bond order.
Correct Answer

verified
Carbon and oxygen form single,double and triple bonds.The C=O bond in carbon monoxide is roughly three times as strong as the C-O single bond,while the C=O bond is about twice as strong as the single bond.The bond energy is approximately proportional to the bond order..
Correct Answer
verified
Multiple Choice
Which of the following contains ionic bonding?
A) CO
B) SrF2
C) Al
D) OCl2
E) HCl
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Oxygen difluoride is an unstable molecule that reacts readily with water.Calculate the bond energy of the O-F bond using the standard enthalpy of reaction and the bond energy data provided.
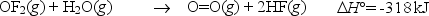

Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitrogen and hydrogen combine to form ammonia in the Haber process.Calculate (in kJ) the standard enthalpy change H° for the reaction written below,using the bond energies given.
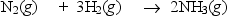 Bond:
N N
H-H
N-H
Bond:
N N
H-H
N-H

A) -969 kJ
B) -204 kJ
C) -105 kJ
D) 204 kJ
E) 595 kJ
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Describe in brief how electronegativity values can be used to predict the percent ionic character of a bond between two atoms.
Correct Answer

verified
Take the electronegativity difference ( E...
E...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Select the correct formula for a compound formed from barium and nitrogen.
A) BaN
B) BaN2
C) Ba2N3
D) Ba2N
E) Ba3N2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 60
Related Exams