Correct Answer

verified
(a)The three structures and th...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
The molecule AX2,where A and X are different elements,will have a dipole moment if the molecule is bent.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory,a molecule with the general formula AX5 will have a ______ molecular shape.
A) tetrahedral
B) trigonal planar
C) trigonal pyramidal
D) trigonal bipyramidal
E) see-saw
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
Using SO2 as an example,describe the sort of experimental data which might suggest that no single Lewis structure is an accurate representation of its bonding.
Correct Answer

verified
In the SO2 molecule,the two sulfur-oxygen...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
According to VSEPR theory,a molecule with the general formula AX3E will have a _____ molecular shape.
A) bent
B) trigonal planar
C) trigonal pyramidal
D) tetrahedral
E) triangular
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has no net dipole moment?
A) N2O
B) NF3
C) H2Se
D) TeO3
E) CH3Cl
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following does the nitrogen atom have a formal charge of -1? 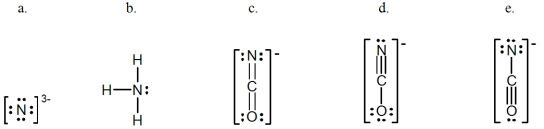
A) a
B) b
C) c
D) d
E) e
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the actual bond angle in SeCl2 using the VSEPR theory.
A) more than 120°
B) between 109° and 120°
C) between 90° and 109°
D) exactly 90°
E) less than 90°
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Draw Lewis structures,showing all valence electrons,for: a.N b.Br- c.O2 d.SO42-
Correct Answer

verified
Correct Answer
verified
Essay
Draw all important resonance structures of the nitrate ion,NO3-
Correct Answer

verified
Correct Answer
verified
True/False
All possible resonance structures contribute equally to the resonance hybrid.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Draw the Lewis structure of XeF4.Use this structure,in conjunction with VSEPR theory,to predict the shape of this molecule.Outline your reasoning.
Correct Answer

verified
The Lewis structure is shown alongside.T...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
What is the molecular shape of ClF2- as predicted by the VSEPR theory?
A) linear
B) bent
C) see-saw
D) T-shaped
E) L-shaped
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the following Lewis structure for ClO3F,chlorine has a formal charge of ____ and an oxidation number of ____. 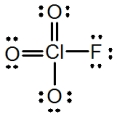
A) 7,7
B) 7,-1
C) 1,1
D) 1,-1
E) 1,7
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
In formaldehyde,CH2O,both the formal charge and the oxidation number of carbon are zero.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The Lewis structure of NO2 violates the octet rule.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the actual bond angles in SF3+ using the VSEPR theory.
A) more than 120°
B) exactly 120°
C) between 109° and 120°
D) between 90° and 109°
E) less than 90°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Phosphoryl iodide is used in the preparation of organophosphorus derivatives and phosphate esters.Select the Lewis structure for POI3 which minimizes formal charges. 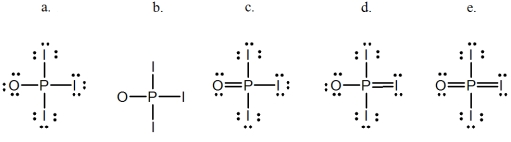
A) a
B) b
C) c
D) d
E) e
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to decide which one of the following ions and molecules is likely to be planar.(The central atom is always first in the formula. )
A) BrF3
B) H3O+
C) PCl3
D) SO42-
E) SF4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory,a molecule with the general formula AX4 will have a ______ molecular shape.
A) bent
B) trigonal planar
C) trigonal pyramidal
D) square planar
E) tetrahedral
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 94
Related Exams