A) 0,-3
B) 0,5
C) 5,-3
D) 5,5
E) 3,5
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory,a molecule with the general formula AX3E2 will have a _____ molecular shape.
A) trigonal pyramidal
B) trigonal bipyramidal
C) trigonal planar
D) T-shaped
E) see-saw
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect? 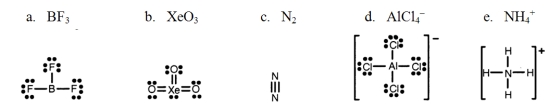
A) a
B) b
C) c
D) d
E) e
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules does not have a dipole moment?
A) CS2
B) H2S
C) CH2Cl2
D) PH3
E) CH2O
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory,a molecule with the general formula AX2E will have a ______ molecular shape.
A) bent
B) see-saw
C) trigonal planar
D) T-shaped
E) trigonal pyramidal
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect? 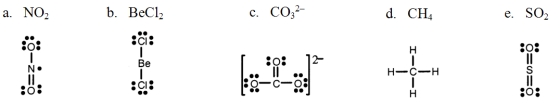
A) a
B) b
C) c
D) d
E) e
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
For the chlorate ion,ClO3-,draw two different valid Lewis structures,as follows: a.a structure in which the octet rule is obeyed b.a structure in which formal charges are minimized
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the actual bond angles in BrF3 using the VSEPR theory.
A) more than 120°
B) exactly 120°
C) between 109° and 120°
D) between 90° and 109°
E) less than 90°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Draw Lewis structures,showing all valence electrons,for the following species: a.S2- b.CO c.SO2 d.CH3OH
Correct Answer

verified
Correct Answer
verified
Essay
Name and outline the concept which is introduced when more than one valid Lewis structure can be drawn for a given molecule or ion.Use appropriate diagrams of the formate ion (HCO2-,carbon is the central atom)to illustrate.
Correct Answer

verified
The concept is resonance.In this situati...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
In order for a non-cyclic triatomic molecule to be bent,VSEPR theory requires that there must be two lone pairs on the central atom.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following is the best Lewis structure a resonance structure? (central atoms are bold)
A) CO2
B) ClO3-
C) COCl2
D) NO2+
E) HCN
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of ClF4- as predicted by the VSEPR theory?
A) square pyramidal
B) square planar
C) see-saw
D) octahedral
E) tetrahedral
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect? 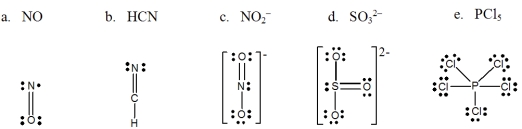
A) a
B) b
C) c
D) d
E) e
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use VSEPR theory to decide which one of the following molecules and ions will have a trigonal pyramidal geometry.(The central atom is always first in the formula. )
A) PCl3
B) BF3
C) SO3
D) BrF3
E) CO32-
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of NOCl as predicted by the VSEPR theory? 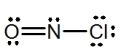
A) linear
B) trigonal planar
C) bent
D) tetrahedral
E) trigonal pyramidal
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of XeO2F2 as predicted by the VSEPR theory? 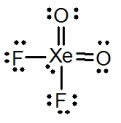
A) square planar
B) tetrahedral
C) square pyramidal
D) see-saw
E) octahedral
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of BeH2 as predicted by the VSEPR theory?
A) linear
B) bent
C) angular
D) trigonal
E) None of these choices is correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In a Lewis structure for a molecule or ion,the sum of the formal charges on the atoms is equal to the charge on the molecule or ion.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles in AsCl3 using the molecular shape given by the VSEPR theory.
A) 90°
B) 109°
C) 120°
D) 180°
E) between 110 and 120°
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 94
Related Exams