A) In the hydrogen atom, the energy of an orbital depends only on the value of the quantum number n.
B) In many-electron atoms the energy of an orbital depends on both n and l.
C) Inner electrons shield outer electrons more effectively than do electrons in the same orbital.
D) The splitting of sublevels in many-electron atoms is explained in terms of the penetration effect.
E) The energy of a given orbital increases as the nuclear charge Z increases.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Atomic size decreases across a period due to an increase in the effective nuclear charge, Zeff.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the largest atomic size?
A) S
B) Ca
C) Ba
D) Po
E) Rn
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
"Electrons added to atomic orbitals of the same energy will remain unpaired with parallel spins until the subshell is more than half-filled" is a statement of
A) the aufbau principle.
B) Hund's rule.
C) the Pauli exclusion principle.
D) the periodic law.
E) the singularity rule.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Elements in the same period of the periodic table have similar valence shell electron configurations.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Moseley's measurements of nuclear charges of the elements provided the basis for arranging the elements of the periodic table in order of increasing atomic number.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the largest first ionization energy?
A) Na
B) Cl
C) Ca
D) Te
E) Br
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
In a single atom, write down the maximum possible number of electrons
a. with n = 3
b. with the designation 4dxy
c. with n = 5, l = 3, ms = + 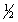
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct electron configuration for Te (Z = 52) .
A) [Kr]5s25p64d8
B) [Kr]5s25d105p4
C) [Kr]5s24d105p6
D) [Kr]5s24f14
E) [Kr]5s24d105p4
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In neutral atoms, the 3d orbitals have higher energy than the 4s orbitals.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the ground state of an atom of silver (Ag) , how many electrons will there be with the quantum number l = 1? (The n, ml, and ms quantum numbers may have any appropriate values.)
A) 9
B) 12
C) 18
D) 24
E) 36
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
According to the exclusion principle, two is the maximum number of electrons in an atom which can share the same four quantum numbers.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elements with the highest first ionization energies are found in the ___________ region of the periodic table.
A) lower left
B) upper left
C) center
D) lower right
E) upper right
G) A) and D)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
Which of the following elements is paramagnetic?
A) Kr
B) Zn
C) Sr
D) V
E) Ar
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Write down the full electron configuration for ground state atoms of a. element number 12. b. element number 23. c. element number 32.
Correct Answer

verified
a. 1s22s22p63s2
b. 1s22s22p63s23p64s23d3
c. 1s22s22p63s23p64s23d104p2
Correct Answer
verified
Multiple Choice
Which of the following electron configurations is impossible?
A) 1s22s22p63s23p4
B) 1s22s22p53s13p4
C) 1s22s22p63s23p63d104s2
D) 1s22s22p63s33p4
E) 1s12s22p63s23p4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following sets of elements have the [noble gas]ns2np2 valence electron configuration?
A) O, S, Se, Te, Po
B) N, P, As, Sb, Bi
C) F, Cl, Br, I, At
D) C, Si, Ge, Sn, Pb
E) Ti, Zr, Hf
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the paramagnetic ion.
A) Cu+
B) Ag+
C) Fe3+
D) Cd2+
E) Ca2+
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most basic oxides are formed from elements found in the __________________ region of the periodic table.
A) upper right
B) upper left
C) center
D) lower right
E) lower left
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has the most negative electron affinity?
A) H
B) Li
C) Na
D) K
E) Rb
G) None of the above
Correct Answer

verified
A
Correct Answer
verified
Showing 1 - 20 of 85
Related Exams