A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the adhesive forces between a liquid and the walls of a capillary tube are greater than the cohesive forces within the liquid,
A) the liquid level in a capillary tube will rise above the surrounding liquid and the surface in the capillary tube will have a convex meniscus.
B) the liquid level in a capillary tube will rise above the surrounding liquid and the surface in the capillary tube will have a concave meniscus.
C) the liquid level in a capillary tube will drop below the surrounding liquid and the surface in the capillary tube will have a convex meniscus.
D) the liquid level in a capillary tube will drop below the surrounding liquid and the surface in the capillary tube will have a concave meniscus.
E) the liquid level in a capillary tube will be flat.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is true?
A) The higher the viscosity,the faster a liquid flows.
B) The viscosity increases with increasing temperature.
C) The stronger the intermolecular forces,the higher the viscosity.
D) Hydrogen bonding in water gives rise to its unusually low viscosity.
E) The viscosity of gases is larger than the viscosity of liquids.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
___________ ___________ is a special type of dipole-dipole interaction.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The energy required to increase the surface of a liquid per unit area is called the
A) capillary action.
B) surface tension.
C) viscosity.
D) cohesion.
E) specific elasticity.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest)type of intermolecular force present in H2S(g).
Correct Answer

verified
Correct Answer
verified
True/False
The maximum number of phases of a single substance which can coexist in equilibrium is two.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metal such as chromium in the body-centered cubic lattice has _______________ atom(s) per unit cell.
A) 1
B) 2
C) 3
D) 4
E) 9
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest)type of intermolecular force present in RbCl(s).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of liquid water in equilibrium with its water vapor in a rigid container at 35ºC.The circles represent water vapor. 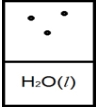 Which diagram below best represents liquid water in equilibrium with its water vapor at 70ºC? The heat of vaporization of water is 40.7 kJ/mol.(R = 8.314 J/K • mol)
Which diagram below best represents liquid water in equilibrium with its water vapor at 70ºC? The heat of vaporization of water is 40.7 kJ/mol.(R = 8.314 J/K • mol)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the pair of substances in which the one with the higher vapor pressure at a given temperature is listed first.
A) C7H16,C5H12
B) CCl4,CBr4
C) H2O,H2S
D) CH3CH2OH,CH3-O-CH3
E) Xe,Kr
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following pure substances will exhibit hydrogen bonding?
A) CH4
B) H2C=CH2
C) H2O
D) CHCl3
E) CHF3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What quantity of heat is required to melt 2.00 kg of iron at its melting point (1809 K) ? For iron, ΔHfus = 13.80 kJ/mol.
A) 0.385 kJ
B) 6.90 kJ
C) 27.6 kJ
D) 494 kJ
E) 771 kJ
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many phase changes are represented in the following heating curve of a pure substance? 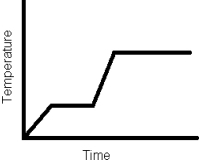
A) 0
B) 1
C) 2
D) 3
E) 4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
In the packing of identical atoms with cubic unit cells,the packing efficiency increases as the coordination number increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diethyl ether,(CH3CH2) 2O,used as a solvent for extraction of organic compounds from aqueous solutions,has a high vapor pressure,which makes it a potential fire hazard in laboratories in which it is used.How much energy is released when 100.0 g is cooled from 53.0°C to 10.0°C? Normal boiling point 34.5°C Heat of vaporization 351 J/g Specific heat of (CH3CH2) 2O(l) 3.74 J/g • °C Specific heat of (CH3CH2) 2O(g) 2.35 J/g • °C
A) 10.1 kJ
B) 16.1 kJ
C) 21.6 kJ
D) 35.1 kJ
E) 48.6 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The arrow in this phase diagram represents which phase change? 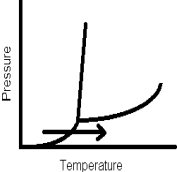
A) sublimation
B) deposition
C) freezing
D) vaporization
E) fusion
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Naphthalene sublimes at room temperature and 1 atmosphere pressure; which statement is true concerning the triple point of naphthalene?
A) The triple point is lower than room temperature.
B) The triple point is higher than room temperature.
C) The triple point is equal to room temperature.
E) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following lacks a regular three-dimensional arrangement of atoms?
A) crystalline solids
B) ionic crystals
C) solids
D) amorphous solids
E) molecular solids
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of the unit cell for nickel(II) oxide.How many O2? anions are there in the unit cell? 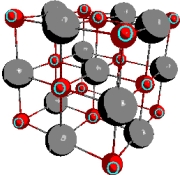
A) 1
B) 4
C) 8
D) 13
E) 14
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 135
Related Exams