A) Adhesion
B) Ionization
C) Cohesion
D) Surface tension
E) Viscosity
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Platinum has a face-centered cubic crystal structure and a density of 21.5 g/cm3.What is the radius of a platinum atom?
A) 69 pm
B) 98 pm
C) 139 pm
D) 196 pm
E) 277 pm
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
Ionic crystals are composed of charged spheres that are held together by covalent bonds.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Ice is less dense than water due to the formation of hydrogen bonds.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct equation for the molar heat of sublimation?
A) ΔHsub = ΔHfus - ΔHvapVliq
B) ΔHsub = ΔHdep + ΔHfus
C) ΔUsub= ΔUfus + ΔUvap
D) ΔHsub = ΔHfus - ΔHvap
E) ΔHsub = ΔHfus + ΔHvap
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the intermolecular force that exists between a magnesium ion and hydrogen sulfide?
A) dipole-dipole forces
B) London dispersion forces
C) ionic bonding
D) ion-dipole forces
E) ion-ion forces
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is true about phase diagrams?
A) Every phase diagram has only one triple point.
B) The slope of the liquid-solid phase boundary line is typically negative.
C) The triple point temperature is the lowest temperature where a liquid can exist.
D) The critical temperature is lower than the triple point temperature.
E) The critical temperature is lower than the normal boiling point.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane is a component of fuel used in internal combustion engines.The dominant intermolecular forces in octane are
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonds.
E) carbon-hydrogen bonds.
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
A face-centered crystal lattice has one atom in the center of the unit cell.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At what temperature does ethanol boil on a day in the mountains when the barometric pressure is 547 mmHg? The heat of vaporization of ethanol is 39.3 kJ/mol and its normal boiling point is 78.3°C.(R = 8.314 J/K • mol)
A) 76.5°C
B) 69.9°C
C) 10.0°C
D) 77.9°C
E) 74.6°C
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pure substances has the strongest dispersion forces?
A) C4H10
B) C5H12
C) C6H14
D) C7H16
E) C8H18
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much energy (heat) is required to convert 52.0 g of ice at -10.0°C to steam at 100°C? Specific heat of ice 2.09 J/g • °C Specific heat of water 4.18 J/g • °C Specific heat of steam 1.84 J/g • °C Molar heat of fusion 6.02 kJ/mol Molar heat of vaporization 40.7 kJ/mol
A) 12.0 kJ
B) 23.0 kJ
C) 46.4 kJ
D) 158 kJ
E) 2.30 ×104 kJ
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Give an example of an ionic crystal and a metallic crystal which contain an element common to each.
Correct Answer

verified
NaCl = ion...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Polyethylene plastic consists of long chains of carbon atoms,each of which is also bonded to hydrogens as shown below: 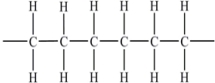 Water forms beads when placed on a polyethylene surface.Why?
Water forms beads when placed on a polyethylene surface.Why?
Correct Answer

verified
Water is highly polar and poly...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
If liquid bromine is cooled to form a solid,which type of solid does it form?
A) atomic
B) metallic
C) molecular
D) ionic
E) covalent
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane has a vapor pressure of 40.torr at 45.1°C and 400.torr at 104.0°C.What is its heat of vaporization? (R = 8.314 J/K • mol)
A) 39.1 kJ/mol
B) 46.0 kJ/mol
C) 590 kJ/mol
D) 710 kJ/mol
E) None of these choices is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of atoms in a body-centered cubic unit cell is
A) 1.
B) 2.
C) 3.
D) 4.
E) 8.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which arrow represents the process of vaporization?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is determined by the magnitude of intermolecular forces in a liquid and is a measure of a fluid's resistance to flow?
A) Surface tension
B) Adhesion
C) Polarity
D) Viscosity
E) Cohesion
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
___________ is the attraction of unlike molecules involved in capillary action.
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 135
Related Exams