A) increases,increases
B) increases,decreases
C) decreases,increases
D) decreases,decreases
E) None of these is generally correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following contains ionic bonding?
A) CO
B) SrF2
C) Al
D) OCl2
E) HCl
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A triple bond cannot exist between a carbon atom and a hydrogen atom.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which response includes all the molecules below that have a central atom that does not follow the octet rule? (1) H2S,(2) BCl3,(3) PH3,(4) SF4
A) (2) and (4)
B) (2) and (3)
C) (1) and (2)
D) (3) and (4)
E) (1) and (4)
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on ionic radii and charges,which of the following solids would have the greatest lattice energy?
A) SrO
B) CsI
C) MgCl2
D) KF
E) NaF
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis dot symbol for the S2- ion is
A) ![]()
B) ![]() 2-
2-
C) S2-
D) ![]()
E) ![]()
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these compounds is most likely to be ionic?
A) KF
B) CCl4
C) CS2
D) CO2
E) ICl
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a covalent compound?
A) Na2O
B) CaCl2
C) Cl2O
D) CsCl
E) Al2O3
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the resonance structure of sulfur dioxide,the sulfur atom has one single bond and one double bond.What is the formal charge of this sulfur atom?
A) 0
B) +1
C) -1
D) +2
E) -2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis structure for a chlorate ion,ClO3-,should show ____ single bond(s) ,____ double bond(s) ,and ____ lone pair(s) .
A) 2,1,10
B) 3,0,9
C) 2,1,8
D) 3,0,10
E) 2,1,9
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an ionic compound?
A) H2S
B) NH3
C) I2
D) KI
E) CCl4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is wrong with this Lewis structure? 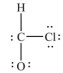
A) There are too many electrons.
B) There are too few electrons.
C) The O atom does not have an octet.
D) The C atom does not have an octet.
E) There is nothing wrong with this Lewis structure.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Lewis dot symbol for the chloride ion is
A) ![]()
B) ![]() -
-
C) ![]() -
-
D) ![]()
E) Cl-
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following species is the central atom (the first atom in the formula) likely to violate the octet rule?
A) BF4-
B) XeO3
C) SiCl4
D) NH3
E) CH2Cl2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many dots does the Lewis dot symbol for argon have around it?
A) 1
B) 2
C) 4
D) 6
E) 8
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 95 of 95
Related Exams